Proteome Profiler Antibody Array for
Tumor Microenvironment (TME)

Tumor microenvironment 연구에 antibody array를 어떻게 사용할 수 있을까요?
Reference 1.
CD276 is an important player in macrophage recruitment into the tumor and an upstream regulator for PAI-1
Scientific Reports volume 11, Article number: 14849 (2021)
DOI https://doi.org/10.1038/s41598-021-94360-9
Abstract
More than 70% of colorectal, prostate, ovarian, pancreatic and breast cancer specimens show expression of CD276 (B7–H3), a potential immune checkpoint family member. Several studies have shown that high CD276 expression in cancer cells correlates with a poor clinical prognosis. This has been associated with the presence of lower tumor infiltrating leukocytes. Among those, tumor-associated macrophages can comprise up to 50% of the tumor mass and are thought to support tumor growth through various mechanisms. However, a lack of information on CD276 function and interaction partner(s) impedes rigorous evaluation of CD276 as a therapeutic target in oncology. Therefore, we aimed to understand the relevance of CD276 in tumor-macrophage interaction by employing a 3D spheroid coculture system with human cells. Our data show a role for tumor-expressed CD276 on the macrophage recruitment into the tumor spheroid, and also in regulation of the extracellular matrix modulator PAI-1. Furthermore, our experiments focusing on macrophage-expressed CD276 suggest that the antibody-dependent CD276 engagement triggers predominantly inhibitory signaling networks in human macrophages
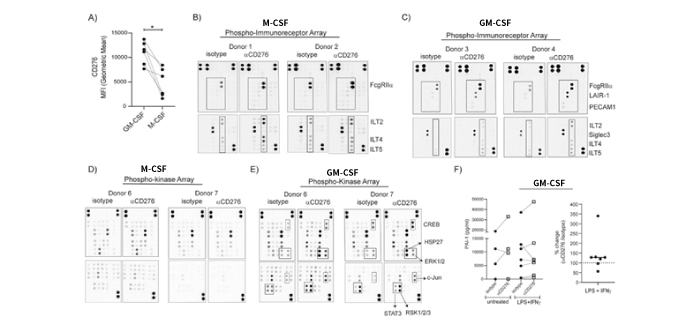
Figure 5. Targeting CD276 on macrophages with specific antibodies induces distinct immunoreceptor phosphorylation accompanied with kinase phosphorylation. (A) Monocytes were cultured either in GM-CSF or M-CSF supplemented medium for 7 days. CD276 expression on monocyte-derived macrophages (MDMs) was analyzed by flow cytometry and CD276 MFI values from six paired samples are summarized in the graph. (B-E) After 7 days culture, MDMs were reseeded for Phospho-Immunoreceptor and Phospho-kinase array. After 3 h resting, MDMs were treated with αCD276 antibody or isotype for 10 min for Phospho-immunoreceptor array and 30 min for Phospho-Kinase array. Cell lysates were processed for phosphorylation signatures. In total macrophages from 4–5 donors were tested. Two representative donors are shown for Phospho-Immunoreceptor arrays: (B) M-CSF MDMs and (C) GM-CSF MDMs. Two representative and matching donors are shown for Phospho-Kinase arrays: (D) M-CSF MDMs and (E) GM-CSF MDMs. (F) MDMs (GM-CSF) were left untreated or treated with LPS + IFNγ in the presence of αCD276 antibody or isotype control for 24 h. The graphs summarize the concentration of PAI-1 in the supernatants. Each dot represents one donor.
Reference 2.
Crosstalk between cancer-associated fibroblasts and immune cells in peritoneal metastasis: inhibition in the migration of M2 macrophages and mast cells by Tranilast
Gastric Cancer (2022), DOI https://doi.org/10.1007/s10120-021-01275-5
Abstract
Background
The role of tumor–stroma interactions in tumor immune microenvironment (TME) is attracting attention. We have previously reported that cancer-associated fibroblasts (CAFs) contribute to the progression of peritoneal metastasis (PM) in gastric cancer (GC), and M2 macrophages and mast cells also contribute to TME of PM. To elucidate the role of CAFs in TME, we established an immunocompetent mouse PM model with fibrosis, which reflects clinical features of TME. However, the involvement of CAFs in the immunosuppressive microenvironment remains unclear. In this study, we investigated the efficacy of Tranilast at modifying this immune tolerance by suppressing CAFs.
Methods
The interaction between mouse myofibroblast cell line LmcMF and mouse GC cell line YTN16 on M2 macrophage migration was investigated, and the inhibitory effect of Tranilast was examined in vitro. Using C57BL/6J mouse PM model established using YTN16 with co-inoculation of LmcMF, TME of resected PM treated with or without Tranilast was analyzed by immunohistochemistry.
Results
The addition of YTN16 cell-conditioned medium to LmcMF cells enhanced CXCL12 expression and stimulated M2 macrophage migration, whereas Tranilast inhibited the migration ability of M2 macrophages by suppressing CXCL12 secretion from LmcMF. In PM model, Tranilast inhibited tumor growth and fibrosis, M2 macrophage, and mast cell infiltration and significantly promoted CD8 + lymphocyte infiltration into the tumor, leading to apoptosis of cancer cells by an immune response.
Conclusion
Tranilast improved the immunosuppressive microenvironment by inhibiting CAF function in a mouse PM model. Tranilast is thus a promising candidate for the treatment of PM.
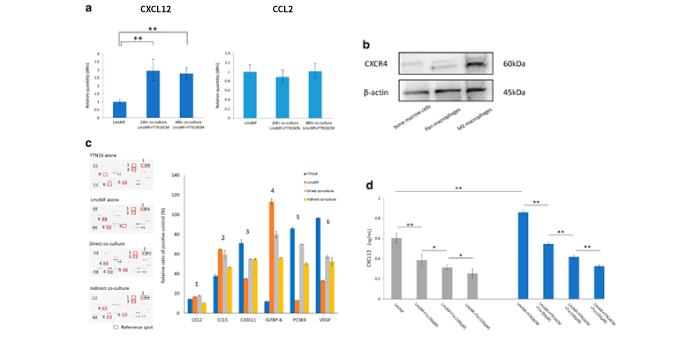
Figure 2. LmcMF cells enhance CXCL12 secretion when the co-culture with YTN16 cell-conditioned medium and Tranilast inhibits this effect. a LmcMF cells were cultured with conditioned medium (CM) of YTN16 cells for 48 h. CXCL12 and CCL2 mRNA expression levels in LmcMF cells cultured with YTN16 cells and CM were analyzed by PCR analysis. b The expression of CXCR4 in mouse bone marrow cells, pan-macrophages (after M-CSF stimulation), and M2 macrophages (after M-CSF and IL-4 stimulation). c Cytokine/chemokine arrays of YTN16 and LmcMF cells and direct/indirect co-culture supernatants: quantitative analysis of the relative levels of 111 factors. Values are normalized to positive control. The bar graphs show the representative chemokine values that changed in expression between samples, but no factors were upregulated by co-culture. d The concentrations of CXCL12 from LmcMF cells were examined by ELISA. Tranilast inhibited the secretion of CXCL12 from LmcMF cells and inhibited the upregulation of CXCL12 by the addition of YTN16 cells-CM in a concentration-dependent manner. Data represent the mean ± SEM of triplicate wells for three independent experiments (*p < 0.05, **p < 0.01). Tra: Tranilas
|
|
사이트명 :(주)웅비메디텍 / 상호:(주)웅비메디텍 |